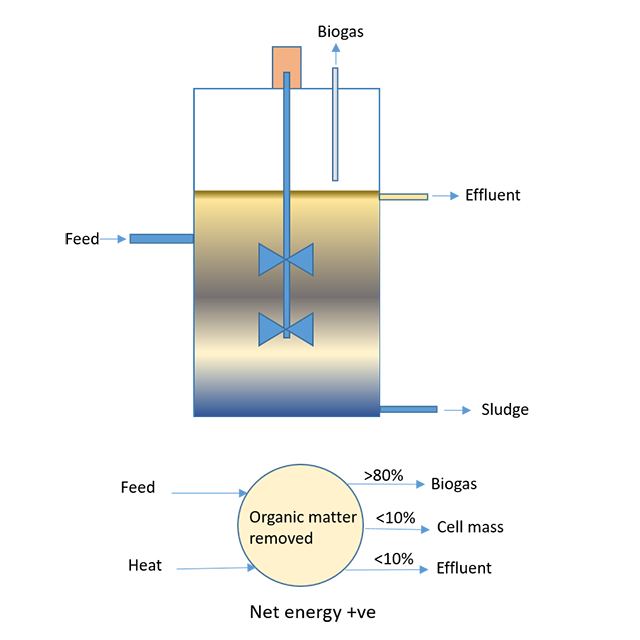
Anaerobic Digestion is Complex, But We Get It!
We provide a wide range of analysis & consultation services to the anerobic digestion (AD) sector. Our multidisciplinary expertise spans feedstock chemistry, biology, process optimsation, and techno-economics. It's the interaction of all these factors that will allow for feedstocks to be efficiently valorised and for the most impactful RNG projects to be developed.
Whether you are just starting out or running a network of biogas plants, our experts can help you reach your goals.

|
|
||||||
We see huge potential for RNG globally. Through innovation, passion, and determination, we at Celignis strive to make a difference in the development of the bioeconomy.
In-Depth Analyses Coupled with Key Insights
We have all the necesary analytical and bioprocess equipment, coupled with the expertise of our team, to undertake comprehensive projects. We then provide detailed reports which also include key summaries of our findings. These allow our clients to make responsive and informed decisions.
Data at Your Fingertips
We know how important your samples are and your need to access data quickly and easily. That's why we created the Celignis Database where you can view results as we get them. So you can view the biogas production curve develop, while BMP digestions are ongoing, rather than having to wait for the analysis to be completed.
Global Recognition as AD/RNG Experts
Celignis provides valued services to over 1000 clients. We understand how the focus of AD projects can differ between countries and have advised a global network of clients on their RNG projects. We also have customs-exemptions for samples sent to us for analysis and research, allowing us to quickly get to work no matter where our clients are based.
We are proud of the knowledge, passion, and work ethic of our team. They have played key roles in the formulation, optimisation, and commercial evaluation of anaerobic digestion processes.

Lalitha Gottumukkala
Founder and Lead of Celignis AD, CIO of Celignis
PhD
Has a deep understanding of all biological and chemical aspects of anaerobic digestion. Has developed Celignis into a renowned provider of AD services to a global network of clients.

Kwame Donkor
AD Services Manager
BSc, MSc, Phd (yr 4)
His PhD focused on optimising AD conditions for Irish feedstocks such as grass. Kwame is now leading the Celignis AD team in the provision of analysis and bioprocess services.

Oscar Bedzo
Technoeconomic Analysis Lead
PhD
A dynamic, purpose-driven chemical engineer with expertise in bioprocess development, process design, simulation and techno-economic analysis over several years in the bioeconomy sector.

Dan Hayes
Celignis CEO and Founder
PhD (Analytical Chemistry)
Dreamer and achiever. Took Celignis from a concept in a research project to being the bioeconomy's premier provider of analytical and bioprocessing expertise.

Piotr Dobkowski
Orders and Data Manager
MAc
Feeds on quality data! Piotr plays a major role in data processing and Orders management at Celignis and is responsible for ensuring AD data are rapidly uploaded to the Celignis Database.

Sajna KV
Bioanalysis Developer
PhD
Our Biomass Detective! Designs, tests, optimizes and validates robust analytical methods for properties of relevance to the anaerobic digestion sector.
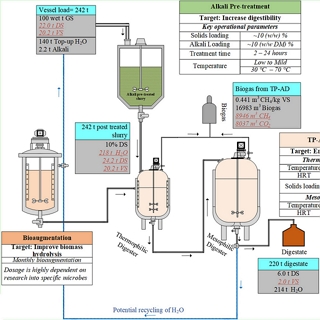
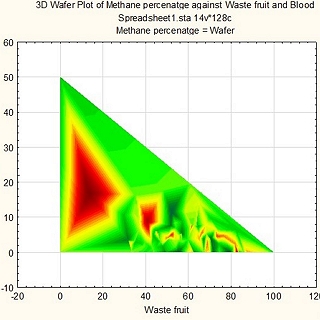
The tool considered seasonality of the locally-available feedstock that could be used as co-feed with the sugar rich waste streams. Also considered, while designing the feedstock mixtures, were Renewable Energy Directive (RED) II GHG emission targets and waste to energy crops ratio.
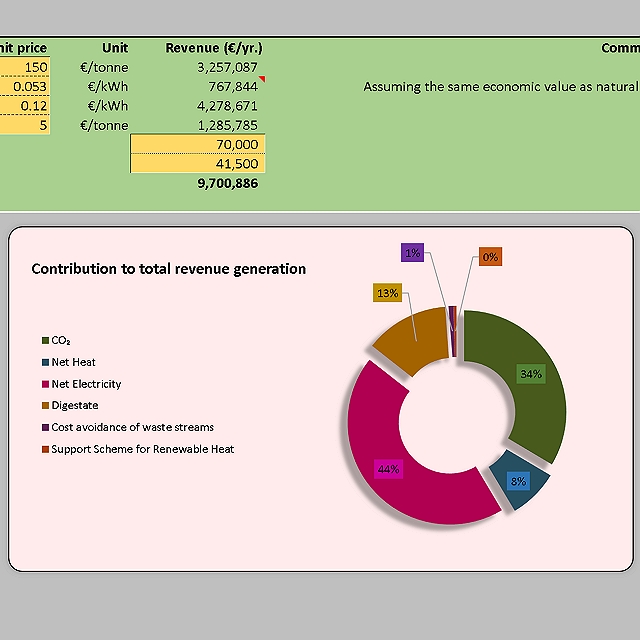
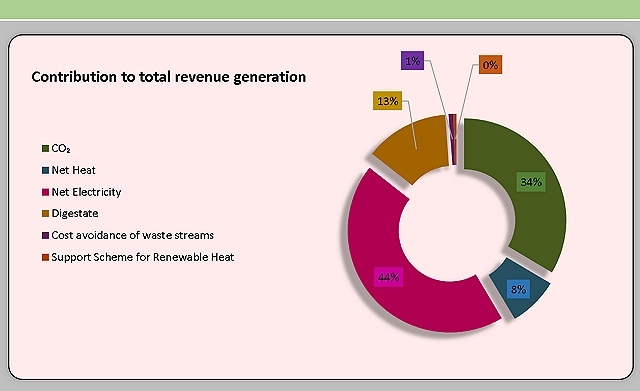
Greenhouse has (GHG) emission reductions and carbon dioxide that could be captured and the total revenue generation from biogas and CO2 were also estimated. The tool allowed the company to make informed decisions on the project and understand the biogas potential and feedstock requirements to meet the target power requirement.
Click here for more information on our technoeconomic analysis (TEA) services for anaerobic digestion and RNG projects, here for detail on our Process Optimisations services, and here for information on our Project Development services.
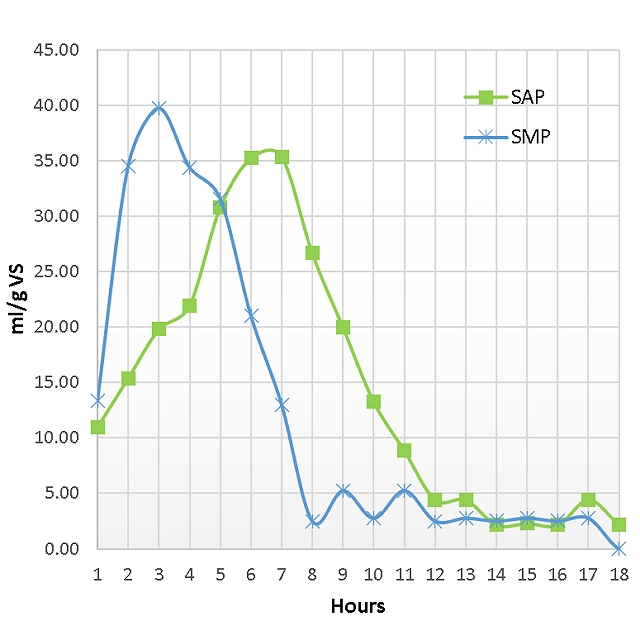
This continuous digestion data, combined with the specific microbial activity tests (Specific Hydrolytic Potential (SHP), Specific Acidogenic Potential (SAP), and Specific Methanogenic Potential (SMP)) on the digestate, provided the plant with the microbial activity in the operational digester and adaptation strategies for the new feedstock.
The full suite of tests and data analysis performed by Celignis allowed the biogas plant operator understand the limitations of the feedstock, feedstock underload/overload effects, optimum feedstock loading, and process indicator (volatile fatty acids (VFAs), alkalinity, biogas production, biogas composition) ranges at different organic loading rates and feedstock mixtures. This allowed adapting the strategies in the biogas plant for maintaining the plant health under feedstock supply and composition variations.
Click here for more information on our Biological Consultation services for the anaerobic digestion/RNG sector, here for information on our Continuious Digestion experiments, and here for information on our Process Optimisation services.
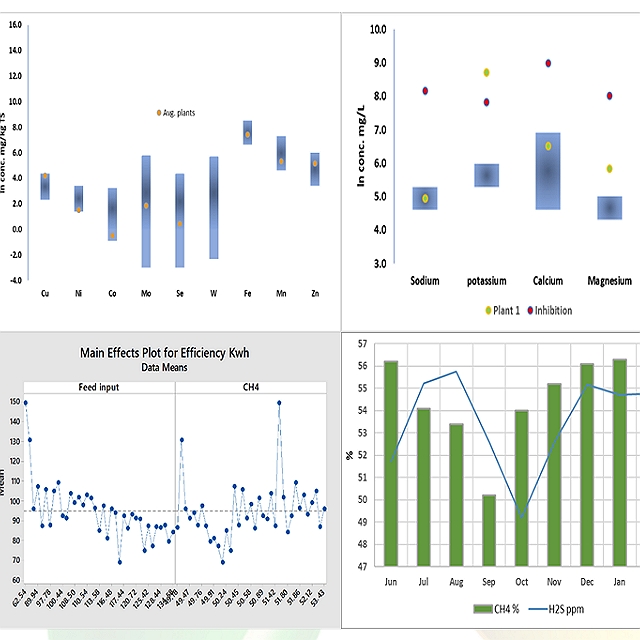
This detailed analysis of the plant process data allowed us to provide operational limits and indicators in the plant beyond common indicators such as VFA and alkalinity and acetic acid to propionic acid ratios (isoforms of volatile fatty acids, presence of traces of hydrogen in the biogas, Hydrogen sulphide and ammonia) and provided green, yellow and red zones for each of the indicators.
In addition to this, Celignis also developed a tool for the company to allow self-design of major and minor elements (nutrients) for the biogas plants based on the feed chemical composition. The tool was designed to be suitable for mono and co-digestion and allows for change from one feedstock to other, and for addition of a new feedstock to the co-digestion mix, without there being a negative affecting on plant performance.
Click here for more information on our Biological Consultation services for the anaerobic digestion/RNG sector, here for information on our services for designing nutrient supplementations, here for information on our Process Optimisation services, and here for information on our analyses of Process Parameters.
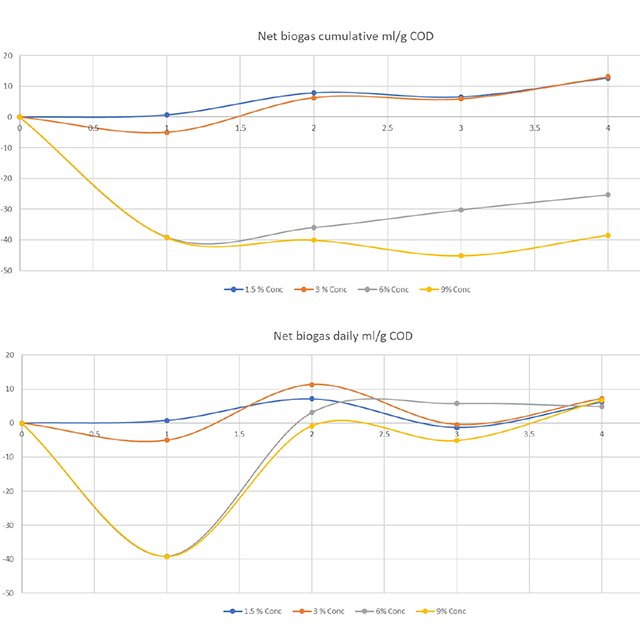
Celignis undertook chemical analyses on the feedstock and then custom-designed and performed Anaerobic Toxicity Assays (ATA) for the waste stream based on the analytical data. These experiments allowed for the determination of threshold feedstock-loadings, as co-feed, in order to avoid the toxic/inhibitory effects of the feedstocks.
The biogas plant is now benefiting from Celignis's support since they can make informed decisions on using feedstocks coming various process industries and so can maintain healthy digestion while incorporating new waste streams into the feed-mix of the RNG plant.
Click here for more information on our Anaerobic Toxicity Assay, and here for information on our Process Optimisation services.