Lab-Scale Experiments for Optimising Bioprocesses
The bioprocess can be a fully vertically-integrated process, involving every stage of processing and conversion, starting from the original feedstock (e.g. corn stover) and ending at the final product (e.g. bioethanol). Alternatively, the bioprocess can be considered to a specific node within a larger sequence of processing steps, for example the pretreatment applied to the corn stover prior to the subsequent hydrolysis and fermentation stages.
Additionally, from the standpoint of downstream processing, which is often one of the most complex and costly stages of a bioprocess, lab-scale experimentation can help identify the most efficient methods for product separation, recovery, and purification.
Background
The Design of Experiments (DoE) approach is a systematic, rigorous procedure used to understand the influence of various factors affecting a process and their interactive effects. It is a statistical tool that aims to optimise processes by not only focusing on the relationship between input parameters and output response but also considering the interdependencies between input variables. The statistical methodology underlying DoE allows for the more efficient and effective exploration of the design space and helps in developing empirical models for process understanding and optimisation.One of the primary advantages of the DoE approach is its efficiency. Rather than conducting numerous experiments that alter one variable at a time, DoE uses a more holistic approach that varies multiple factors simultaneously. This enables researchers to understand how factors interact with each other to influence the process, yielding more comprehensive insights into the process. Moreover, it reduces the number of experiments needed, saving time, effort, and resources.
Another advantage of the DoE approach is its ability to identify optimal process conditions. By systematically varying the factors and examining the process output, DoE can identify the conditions that yield the best result. For a bioprocess this could mean maximizing product yield, purity, or overall process efficiency.
Methodology
The implementation of DoE in bioprocess optimisation at the lab-scale involves a sequence of steps. First, the process parameters (factors) and their potential ranges are identified. Then, an experimental design is chosen. This can be a full-factorial design, where all possible combinations of factors are tested, or a fractional factorial design, where only a subset of the possible combinations is tested. After performing the experiments, the data is analysed, often using regression analysis, to develop an empirical model of the process.In some cases several sequential DoE can be undertaken in order to fully optimise a bioprocesses. For example, there may initially be an uncertainty with regards to which type of pretreatment technology would be most suitable for valorising a particular feedstock. Simply testing each of the candidate pretreatment processes once, using only one set of conditions, may not give the correct answer with regards to which pretreatment is the most suitable since the selected conditions may not be appropriate for that feedstock. Hence, an initial DoE could involve testing a number of different pretreatments with each type of pretreatment being tested over a number of different conditions.
The outputs of these experiments would provide crucial data with regards to the dynamics and efficiency of biomass processing for each pretreatment over a range of conditions. Such data may then be sufficient to allow for a final decision to be made with regards to the selected pretreatment technology. There could then follow a second DoE where, informed by the results from the prior experiments, a more detailed investigation and optimisation of the chosen pretreatment can be undertaken. A further DoE may be possible in some cases and would involve fine-tuning the optimal conditions, based on the relationships elucidated in the prior experiments.
Factors such as mass and heat transfer, mixing, and the behavior of materials often change with scale, and these changes can influence the process performance. alidation at higher TRLs ensures that these factors are taken into account, thereby ensuring the robustness and reliability of the process.
In bioprocess development the general approach is that, after optimising the process at the lab-scale (i.e TRL 3/ 4), the process is then validated at pilot-scale TRLs (e.g. TRL5 or TRL6).
Celignis is able to develop, validate, and optimise bioprocesses at these technology readiness levels. Click below to read more about our facilities and services for pilot-scale bioprocesses.
Get more info...Pilot-Scale
1. Understanding Your Requirements
Prior to undertaking bioprocess projects we learn from our clients what their targets are from the process as well as whether there are any restrictions or requirements that may need to form the boundaries of the work that we undertake. These help to guide us to then prepare a potential bioprocess development project.
2. Detailed Feedstock Analysis
In cases where you have already selected a feedstock for the bioprocess, we would then undertake a detailed compositional analysis (P10 or, ideally, P19) of representative samples of that feedstock.
In cases where the feedstock has not yet been selected we can review your list of candidate feedstocks, selecting top candidates based on our prior experience in their analysis and bioprocessing. If you do not have a list of candidate feedstocks then we can provide one, based on your location and the requirements outlined in Stage 1. We would then analyse in detail these priority feedstocks and come to a decision, based on the compositional data and other relevant factors (e.g. price, supply, consistency etc.) on a selected feedstock for the project.
3. Formulation of DoE
At this point of the project, the Celignis Bioprocess team typically meet to discuss and prepare a project proposal for the development of the bioprocess. This will involve us defining the number and scope of lab-scale optimisation experiments, formulated according to our chosen Design of Experiments (DoE).
It is possible that the work may involve several different experimental datasets, either focused on different stages of the bioprocess (e.g. pretreatment, primary-conversion, product recovery etc.) or on iterative improvements/refinements based on prior experiments. In the former case it is possible that these different sets of experiments could be undertaken in parallel (in order to achieve the project's objectives more quickly) while, in the latter case, the next set of experiments would need to follow the prior set, as the information learnt from earlier work would be needed to set the specific conditions for the follow-up work.
After this proposal is reviewed by the client, and revised if needed, we are then ready to start the lab-work.
4. Undertake Experiments
This stage of the project will involve us undertaking the lab-scale experimental work agreed in Stage 3.
It is possible for the work in this Stage to be phase-gated where the experimental work is broken-up into smaller subsections which, once completed, lead to the provision of reports/deliverables to the clients providing an update on the results and observations. Once a particular phase-gate is completed, in accordance with the requirements and expectations outlined in Stage 3, then we can proceed to work on the next phase.
The division of projects in this manner allows for them to be managed and evaluated more effectively and gives ample opportunities for our clients to provide feedback.
Stage 4 of the project will be completed once the DoE, formulated in Stage 3, has been completed and the final reports issued.
5. Validation at Higher TRLs
This is an optional Stage of the bioprocess development project. It involves the validation of the optimal process conditions, determined in Stage 4 at the lab-scale, at higher technology readiness levels (TRLs). The scales at which we can operate are dependent on the type of technology employed, but can reach up to 100 litres.
We have all of the necessary downstream equipment to efficiently handle the solid and liquid streams arising from these scaled-up activities.
If we find that there are differences between the yield and compositions of the different streams, compared with our lab-scale experiments, then we can explore the potential reasons for these and work on final tweaks to optimise the bioprocess for higher TRLs.
6. Technoeconomic Analysis (TEA)
This is also an optional Stage of the bioprocess development. It involves the Celignis team, including Oscar our chief TEA expert, undertaking a detailed technoeconomic analysis of the developed process. We apply accurate and realistic costing models to determine the CAPEX and OPEX of simulated and pilot scale processes which are then used to determine key economic indicators such as IRR, NPV and payback periods.
Within these TEAs we can undertake sensitivity analyses to assess the effect of variable costs and revenues on the commercial viability of the process.
Our preferred approach is to include TEA studies at each stage of the development of the bioprocess, so that the process can be optimised in a commercially-relevant way, followed by a more detailed TEA after the process has been optimised and tested at higher TRL levels.
Click here to read more about the technoeconomic analysis (TEA) services offered by Celignis.

Lalitha Gottumukkala
Founder of Celignis Bioprocess, CIO of Celignis
PhD
<p style="text-align: left;">Has a deep understanding of all biological and chemical aspects of bioproceses. Has developed Celignis into a renowned provider of bioprocess development services to a global network of clients.</p>

Oscar Bedzo
Bioprocess Project Manager & Technoeconomic Analysis Lead
PhD
<p style="text-align: left;">A dynamic, purpose-driven chemical engineer with expertise in bioprocess development, process design, simulation and techno-economic analysis over several years in the bioeconomy sector.</p>

Dan Hayes
Celignis CEO And Founder
PhD (Analytical Chemistry)
<p style="text-align: left;">Dreamer and achiever. Took Celignis from a concept in a research project to being the bioeconomy's premier provider of analytical and bioprocessing expertise.</p>
Global Recognition as Bioprocess Experts
Extraction
Pretreatment
Hydrolysis
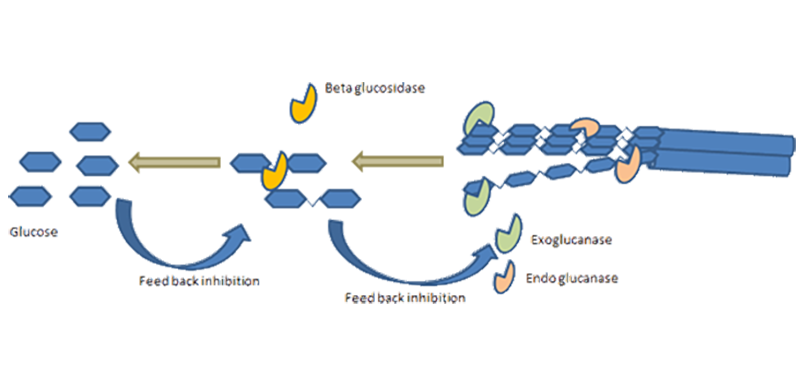
Enzymes
Fermentation
Downstream Processing
TRL Scale-Up
Technoeconomic Analyses
Biobased Chemicals
From Process Refinements to an Entire New Process
Research Collaborations
Hydrothermal carbonization (HTC) research has mainly focused on primary char production, with limited attention to secondary char, which is formed through polymerization and condensation of dissolved organic compounds in the liquid phase. This research aims to address this gap via an experimental investigation of the impact of stirring on the mass and carbon balance of HTC reaction products, surface functional groups, and surface morphology of secondary char, using fructose as a model compound. A 3D hydrodynamic simulation model was developed for a two-liter HTC stirred reactor. The experimental results indicated that stirring did not significantly influence the pH, mass, carbon balance, and surface functional groups of secondary char produced under the range of experimental conditions (180 C, 10% biomass to water (B/W) ratio, and a residence time of 0-120 min) studied. Nonetheless, it was observed that a stirring rate of 200 rpm influenced the morphology and shape of the secondary char microspheres, leading to a significant increase in their size i.e., from 1-2 um in unstirred conditions compared with 70 um at a stirring rate of 200 rpm. This increase in size was attributed to the aggregation of microspheres into irregular aggregates at stirring rates > 65 rpm and residence times > 1 h. The hydrodynamic model revealed that high turbulence of Re > 104 and velocities > 0.17 m s-1 correlated with regions of secondary char formation, emphasizing their role in particle aggregation. Particle aggregation is significant above a stirring rate of 65 rpm, which corresponds to the onset of turbulent flow in the reactor. Finally, a mechanism is proposed, based on reactor hydrodynamics under stirred conditions, that explains secondary char deposition on the reactor walls and stirrer. | |
A dried dairy processing sludge (sludge from wastewater treatment of an effluent from a milk processing plant) was pyrolysed in a single-particle reactor at different temperatures from 400 C to 900 C. NH3 and HCN were measured online and offline by means of FTIR as well as by cumulative sampling in impinger bottles (in 0.05 M H2SO4 and 1 M NaOH, respectively) and analysed by photometric method. NO and NO2 were measured online using a nitric oxide analyser while N2O was measured by FTIR. Nitrogen (N) in the sludge and in the remaining char, char-N, was determined. Moreover, tar content in pyrolysis gas was measured and tar-N was determined. The results with respect to N mass balance closure are discussed. The different measurements techniques are compared. For pyrolysis at 520 and 700 nitrogen in the gas phase was mainly contained as N2 (36 % and 40 % respectively), followed by NH3 (15 % and 18 %), tar-N (10 % and 9 %), HCN (1 % and 3 %), NO (1 %) and NO2 (0.2 %). The dairy processing sludge has very specific properties with organic-N present predominantly as proteins and a high content of inherent Ca. These characteristics affected the distribution of N. The amount of char-N was higher while the amount of tar-N lower than for sewage sludge from literature, at comparable pyrolysis temperature. | |
Dairy processing sludge (DPS) is a byproduct generated in wastewater treatment plants located in dairy (milk) processing companies (waste activated sludge). DPS presents challenges in terms of its management (as biosolids) due to its high moisture content, prolonged storage required, uncontrolled nutrient loss and accumulation of certain substances in soil in the proximity of dairy companies. This study investigates the potential of hydrothermal carbonization (HTC) for recovery of nutrients in the form of solid hydrochar (biochar) produced from DPS originating from four different dairy processing companies. The HTC tests were carried out at 160 C, 180 C, 200 C and 220 C, and a residence time of 1h. The elemental properties of hydrochars (biochars), the content of primary and secondary nutrients, as well as contaminants were examined. The transformation of phosphorus in DPS during HTC was investigated. The fraction of plant available phosphorus was determined. The properties of hydrochar (biochar) were compared against the European Union Fertilizing Products Regulation. The findings of this study demonstrate that the content of nutrient in hydrochars (biochars) meet the requirements for organo-mineral fertilizer with nitrogen and phosphorus as the declared nutrients (13.9-26.7%). Further research on plant growth and field tests are needed to fully assess the agronomic potential of HTC hydrochar (biochar). | |
Disposal of waste-activated sludge [dairy processing sludge, (DPS)] from wastewater treatment plants located in milk processing companies is an increasing concern. DPS is usually applied to farmlands in the vicinity of the dairy companies. This practice is becoming unsustainable due to uncontrolled nutrient loss and potential soil contamination. We propose to recover nutrients in the form of biochar. This paper examines the properties of biochars obtained from slow pyrolysis of DPS. DPS samples were pyrolyzed at laboratory and pilot scale at 600 and 700 C. The elemental properties of biochars, the content of primary and secondary nutrients, as well as contaminants were examined and compared against the European Union Fertilizing Products Regulation. The biochars meet the specified limits for hydrogen-to-organic carbon ratio, chloride, and polycyclic aromatic hydrocarbons intended for gasification and pyrolysis component category materials. In six out of eight biochars, the content of phosphorus (P) as a single declared nutrient and the level of contaminants meet those required for an organo-mineral fertilizer. Only two biochars meet the required concentrations of nitrogen, phosphorus, and potassium. A minimum solid content of 30% in DPS is required to make the process of biochar production energetically sustainable. | |
Anaerobically digested sewage sludge mixed with forest residues was pyrolysed at 800 C, at laboratory and pilot scale. The study quantified differences in char and gas yields for tests carried out in a simple fixed bed laboratory reactor and rotating retort pyrolyser at pilot scale, when the residence time of feedstock was 10 min in both cases. The yield of char from pilot scale was 4 % lower than from laboratory scale while the yield of gas was 15.7 % higher. During the pilot scale pyrolysis of anaerobically digested sewage sludge blended with forest residues the gas quality for energy recovery applications was assessed and the fate of impurities (tar, NH3 and H2S) was investigated. The raw pyrolysis gas contained 14.6 g/Nm3 of tar, 36.9 g/Nm3 of NH3 and 793 ppm of H2S. Sixteen N-containing tar species were identified of which pyridine, propenenitrile, 2-methyl-, benzonitrile, and indole are found to be the most abundant. The yield of N-containing tar compounds accounted for approx. 12 % of total tar content. Conditioned pyrolysis gas contained 7.1 g/Nm3 of tar, 0.036 g/Nm3 of NH3 and 119 ppm of H2S. Benzene was by far the most abundant tar compound followed by toluene and styrene. The specifications of the used internal combustion engine were exceeded due to the sum of tar compounds such as fluorantrene and pyrene with 4+ aromatic rings (at 0.0015 g/Nm3) and NH3 content The effectiveness and sustainability of energy recovery in wastewater treatment can be improved using forest industry by-products. | |
Adsorption of six contaminants of emerging concern (CECs) - caffeine, chloramphenicol, carbamazepine, bisphenol A, diclofenac, and triclosan - from a multicomponent solution was studied using activated biochars obtained from three lignocellulosic feedstocks: wheat straw, softwood, and peach stones. Structural parameters related to the porosity and ash content of activated biochar and the hydrophobic properties of the CECs were found to influence the adsorption efficiency. For straw and softwood biochar, activation resulted in a more developed mesoporosity, whereas activation of peach stone biochar increased only the microporosity. The most hydrophilic CECs studied, caffeine and chloramphenicol, displayed the highest adsorption (22.8 and 11.3 mg g-1) onto activated wheat straw biochar which had the highest ash content of the studied adsorbents (20 wt%). Adsorption of bisphenol A and triclosan, both relatively hydrophobic substances, was highest (31.6 and 30.2 mg g-1) onto activated biochar from softwood, which displayed a well-developed mesoporosity and low ash content. | |
Magnetic carbons can significantly lower the costs of wastewater treatment due to easy separation of the adsorbent. However, current production techniques often involve the use of chlorinated or sulfonated Fe precursors with an inherent potential for secondary pollution. In this study, ochre, an iron-rich waste stream was investigated as a sustainable Fe source to produce magnetic activated biochar from two agricultural feedstocks, softwood and wheat straw. Fe doping resulted in significant shifts in pyrolysis yield distribution with increased gas yields (+50%) and gas energy content (+40%) lowering the energy costs for production. Physical activation transformed ochre to magnetite/maghemite resulting in activated magnetic biochars and led to a 4-fold increase in the adsorption capacities for two common micropollutants - caffeine and fluconazole. The results show that Fe doping not only benefits the adsorbent properties but also the production process, leading the way to sustainable carbon adsorbents. | |
The majority of the sludge from the treatment of wastewater in milk processing plants is land spread. The drawbacks of land spreading include local oversupply due to high transport costs, which results in sludge being spread on lands in the vicinity of the dairy factories. Local oversupply can lead to accumulation of certain substances in soil through annual application over many years. Therefore, in the long term, there is a need for alternative methods to recover energy and nutrients from increasing volumes of sludge generated from dairy processing. Pyrolysis offers a potential alternative to land spreading, which can reduce health and environmental risks, while providing an avenue for the recovery of energy and nutrients. Pyrolysis allows energy recovery in the form of a high calorific value pyrolysis gas and a char which may be used as a soil amendment. In this study pyrolysis of dried dairy sludge was carried out at pilot scale. The results indicate that a dried biological sludge can be successfully pyrolysed and when mixed with wood the resulting char meets European Biochar Certificate criteria regarding carbon content. Most of the initial energy content of the feedstock was retained in the pyrolysis gas prior to cleaning, 53%, compared to 34.5% in the char and 1.5% in the tar. For the pyrolysis gas after cleaning (mainly cracking in presence of air) the initial energy content of the feedstock retained in the gas was only slightly higher than that retained in the char, 39.2% versus 34.5%, while the tar accounted for 0.8% of the initial energy content. | |
Eucalypts can be very productive when intensively grown as short rotation woody crops (SRWC) for bioproducts. In Florida, USA, a fertilized, herbicided, and irrigated cultivar planted at 2471 trees/ha could produce over 58 green mt/ha/year in 3.7 years, and at 2071 trees/ha, its net present value (NPV) exceeded $750/ha at a 6% discount rate and stumpage price of $11.02/green mt. The same cultivar grown less intensively at three planting densities had the highest stand basal area at the highest density through 41 months, although individual tree diameter at breast height (DBH) was the smallest. In combination with an organic fertilizer, biochar improved soil properties, tree leaf nutrients, and tree growth within 11 months of application. Biochar produced from Eucalyptus and other species is a useful soil amendment that, especially in combination with an organic fertilizer, could improve soil physical and chemical properties and increase nutrient availability to enhance Eucalyptus tree nutrition and growth on soils. Eucalypts produce numerous naturally occurring bioproducts and are suitable feedstocks for many other biochemically or thermochemically derived bioproducts that could enhance the value of SRWCs. | |