Biomass Chemistry
A good understanding of biomass chemistry is crucial in selecting and optimising suitable biomass conversion technologies for a given feedstock.
For example, for the production of biobased chemicals and advanced biofuels the chemical composition of a sample will determine whether
it should be processed in hydrolysis or thermochemical processes and which types of
pre-treatment technology is most suitable.
For hydrolysis technoloies (e.g. enzymatic hydrolysis, acid hydrolysis) the lignocellulosic (cellulose, hemicellulose, lignin) constituents of a sample are critical whilst for thermochemical technologies the elemental and ash composition of the sample are of greater importance.
Celignis is home to a multidisciplinary team of biomass experts with an in-depth understanding of the diverse chemistry of biomass and its relevance to valorisation approaches. Below we wil guide you through some of the main constituents of biomass. We are also available to discuss any particular questions you may have regarding how to best use a given feedstock or how to select the most suitable biomass for your conversion process.
For hydrolysis technoloies (e.g. enzymatic hydrolysis, acid hydrolysis) the lignocellulosic (cellulose, hemicellulose, lignin) constituents of a sample are critical whilst for thermochemical technologies the elemental and ash composition of the sample are of greater importance.
Celignis is home to a multidisciplinary team of biomass experts with an in-depth understanding of the diverse chemistry of biomass and its relevance to valorisation approaches. Below we wil guide you through some of the main constituents of biomass. We are also available to discuss any particular questions you may have regarding how to best use a given feedstock or how to select the most suitable biomass for your conversion process.
Lignocellulosic biomass is that which predominately contains the polysaccharides cellulose and hemicellulose and the biopolymer lignin. There will also be lesser amounts of ash and
extractive components.
Click here to see the Celignis Analysis Packages related to the characterisation of lignocellulosic samples.
Click here to see the Celignis Analysis Packages related to the characterisation of lignocellulosic samples.
Cellulose is the major mass constituent of most naturally occurring biomass. It is a polysaccharide (a polymer containing sugar units) made up of
interlinked glucose units. These bonds are beta-linked and result in significant hydrogen bonding,
both within and between cellulose molecules, meaning that cellulose is relatively hard to hyrolyse (compared with the alpha-linked polymers
in starch).
Cellulose tends to consist of amorphous regions and crystalline regions. The amorphous regions tends to be easily hydrolysable by acids while crystalline
cellulose is more resistant. The result is that hydrolysis with a dilute acid tends to remove the amorphous regions from cellulose leaving a polysaccharide of increased
crystallinity that is resistant to further hydrolysis.
Request a QuoteCellulose
Hemicelluloses constitute the cell wall polysaccharides of land plants that are not cellulose or pectins. Hemicelluloses tend to be heteropolysaccharides
(i.e. containing more than one type of sugar unit) with branches attached to the main backbone.
Hemicelluloses are mostly built up of: the pentoses D-xylose and L-arabinose; the hexoses D-glucose, D-mannose and D-galactose; with smaller amounts of L-rhamnose, in addition to uronic acids such as glucuronic acid, 4-O-methyl-D-glucuronic acid, and galacturonic acid. While the proportions of these substituents vary between hemicellulose and feedstock types, the majority tend to be pentoses. Click here to read more about uronic acids.
Hemicelluloses are mostly built up of: the pentoses D-xylose and L-arabinose; the hexoses D-glucose, D-mannose and D-galactose; with smaller amounts of L-rhamnose, in addition to uronic acids such as glucuronic acid, 4-O-methyl-D-glucuronic acid, and galacturonic acid. While the proportions of these substituents vary between hemicellulose and feedstock types, the majority tend to be pentoses. Click here to read more about uronic acids.
There are key differences between cellulose and hemicelluloses. These include the absence of a highly ordered state in hemicelluloses, and that their molecular weight is
much lower; their degree of poymerisation is only 100 to 200 units (compared to 1000's in cellulose). Such factors help explain their comparatively easy hydrolysis.
However, many cellulases designed for enzymatic hydrolysis of cellulose would not be active in hemicellulose hydrolysis meaning that specific enzymes would be needed if the hemicelluloses are not hydrolysed in the pretreatment steps. Also, hemicelluloses contains sugars other than glucose, including C5 sugars, and these tend to require different biota to those used for glucose fermentation. Each of these sugars may also have different fermentation efficiencies.
However, many cellulases designed for enzymatic hydrolysis of cellulose would not be active in hemicellulose hydrolysis meaning that specific enzymes would be needed if the hemicelluloses are not hydrolysed in the pretreatment steps. Also, hemicelluloses contains sugars other than glucose, including C5 sugars, and these tend to require different biota to those used for glucose fermentation. Each of these sugars may also have different fermentation efficiencies.
Request a QuoteHemicellulose
Lignin is a structurally important polymer in biomass. It is said to function primarily as a supporting agent in cell structure and it
also assists in the resistance of biomass against microbial attack and decay.
Structurally, lignin can be described as a complex three-dimensional polymer of phenylpropane units (approx. 25). The phenylpropane units are derelatives of carbohydrates, coming from the dehydration and cyclisation of sugars. They are mostly either 4-hydroxycinnamyl alcohol (para-coumaryl alcohol, H) or its 3- and/or 3,5-methoxylated derivatives - coniferyl (guaiacyl, G) alcohol, and sinapyl alcohol (or syringal, S), respectively.
Structurally, lignin can be described as a complex three-dimensional polymer of phenylpropane units (approx. 25). The phenylpropane units are derelatives of carbohydrates, coming from the dehydration and cyclisation of sugars. They are mostly either 4-hydroxycinnamyl alcohol (para-coumaryl alcohol, H) or its 3- and/or 3,5-methoxylated derivatives - coniferyl (guaiacyl, G) alcohol, and sinapyl alcohol (or syringal, S), respectively.

The ratio of these units varies between plants; for example in hardwoods S and G forms dominate, whereas softwood lignins
contain only G units. Ether linkages between the phenylpropane units are very resistant to cleavage, a factor in explaining the low lignin degradation rates by most biota.
It has been shown that a high lignin content is correlated with the recalcitrance of polysaccharides to enzymatic hydrolysis and that increasing the relative proportion of the
S monomer may aid in biofuel conversion mechanisms.
However, many cellulases designed for enzymatic hydrolysis of cellulose would not be active in hemicellulose hydrolysis meaning that specific enzymes would be needed if the hemicelluloses are not hydrolysed in the pretreatment steps. Also, hemicelluloses contains sugars other than glucose, including C5 sugars, and these tend to require different biota to those used for glucose fermentation. Each of these sugars may also have different fermentation efficiencies.
A small fraction of lignin is soluble under acid conditions. The amount of acid soluble lignin (ASL) varies between lignin and feedstock types and its presence can interfere with enzymatic hydrolysis and the acid-catalysed hydrolysis of cellulose.
However, many cellulases designed for enzymatic hydrolysis of cellulose would not be active in hemicellulose hydrolysis meaning that specific enzymes would be needed if the hemicelluloses are not hydrolysed in the pretreatment steps. Also, hemicelluloses contains sugars other than glucose, including C5 sugars, and these tend to require different biota to those used for glucose fermentation. Each of these sugars may also have different fermentation efficiencies.
A small fraction of lignin is soluble under acid conditions. The amount of acid soluble lignin (ASL) varies between lignin and feedstock types and its presence can interfere with enzymatic hydrolysis and the acid-catalysed hydrolysis of cellulose.
Request a QuoteLignin
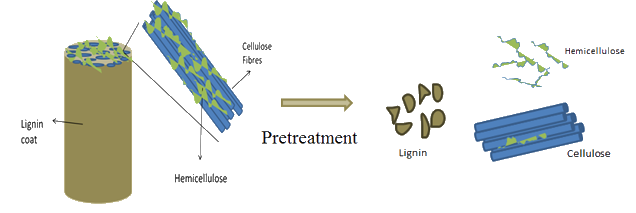
The complex associations of lignin with the polysaccharides is of particular importance for many hydrolysis technologies.
Cellulose is said to form a skeleton which is surrounded by other substances functioning as matrix (hemicelluloses) and encrusting (lignin) materials.
More specifically, disordered (amorphous) cellulose molecules, as well as hemicelluloses and lignin, are located in the spaces between crystalline cellulose microfibrils. The hemicelluloses are considered to be amorphous even though they are apparently orientated in the same direction as the cellulose microfibrils.
More specifically, disordered (amorphous) cellulose molecules, as well as hemicelluloses and lignin, are located in the spaces between crystalline cellulose microfibrils. The hemicelluloses are considered to be amorphous even though they are apparently orientated in the same direction as the cellulose microfibrils.
Researchers have postulated the effects of cross-linking
on the properties of plant cell walls, such as accessibility and degradability. The resistance of polysaccharide-lignin bonds to hydrolysis not only depends on the type of linkage involved but
also on the types of sugars associated with the linkage, and the chemical structure of the lignin unit attached to the sugar.
For the cost-effective production of second generation biofuels in high yields it is important that a pre-treatment is employed to break apart the lignocellulose structure, enabling easier hydrolysis of cellulose. At Celignis we have extensive experience in the analysis of pre-treated biomass samples and have characterised biomass pre-treated by several different processes under varying degrees of severity.
For the cost-effective production of second generation biofuels in high yields it is important that a pre-treatment is employed to break apart the lignocellulose structure, enabling easier hydrolysis of cellulose. At Celignis we have extensive experience in the analysis of pre-treated biomass samples and have characterised biomass pre-treated by several different processes under varying degrees of severity.
Background
Extractives are defined as extraneous components that may be separated from the insoluble cell wall material by their solubility in water or
neutral organic solvents. Solvents of different polarities are required to remove different types of extractives.
Hence the extractives are often classified according to which solvent can extract them (e.g. ethanol-soluble-extractives).
There are a large number of different extractives, many of which are species-specific. These are often classified into categories according
to the similarity of their chemical structures. Major categories include monosaccharides, polysaccharides, volatile oils, terpenes, fatty acids and their esters, waxes, polyhydric alcohols, alkaloids, and aromatic compounds.
Relevance
For lignocellulosic compositional analysis it is very important to remove the extractives prior to hydrolysing the sample with acid.
If the extractives are not removed they will cross-react with the acid and condense to acid insoluble components that will be associated-with and
classified-as Klason lignin (KL). The presence of these extractives during the acid hydrolysis process may also
impact on the hydrolysis dynamics and ultimate fate of the monosaccharides that are liberated.
We have found that hydrolysis with no extraction significantly increases the reported value for KL and acid soluble lignin (ASL). Water and/or 95% ethanol can be effective in removing a large proportion of the extractives, allowing more accurate lignocellulosic analysis. For this reason we typically recommend that analysis package P10, which involves an exhaustive extraction of water followed by 95% ethanol prior to the lignocellulosic analysis of the remaining solid residue, is undertaken for most biomass samples.
Extraction may also be necessary for the solid samples obtained from pre-treatment and other biomass conversion processes in cases where the liquid phase may still be sorbed onto the solid biomass. Click here to read more about our analysis methods for pre-treated samples.
We have found that hydrolysis with no extraction significantly increases the reported value for KL and acid soluble lignin (ASL). Water and/or 95% ethanol can be effective in removing a large proportion of the extractives, allowing more accurate lignocellulosic analysis. For this reason we typically recommend that analysis package P10, which involves an exhaustive extraction of water followed by 95% ethanol prior to the lignocellulosic analysis of the remaining solid residue, is undertaken for most biomass samples.
Extraction may also be necessary for the solid samples obtained from pre-treatment and other biomass conversion processes in cases where the liquid phase may still be sorbed onto the solid biomass. Click here to read more about our analysis methods for pre-treated samples.
However, extractives are not necessarily a complication in biomass valorisation. In some cases there may be high-value constituents in biomass extracts. For example,
in our BBI project UNRAVEL one of Celignis's roles is to characterise the extractives in wood residues and consider methods for the
purification and recovery of any commercially-relevant chemicals in these extracts. At Celignis we have six pressurised liquid extraction units (Dionex Accelerated Solvent Extractors)
suitable for extracting biomass using a wide variety of solvents and conditions. We have also developed a range of analytical protocols for analysing these extracts. Feel free
to get in touch with us if you would like us to evaluate your feedstocks for their extractives composition and click here
if you would like to read more about our methods for analysing extractives.

Request a QuoteExtractives
Click here to see the Celignis Analysis Packages related to the characterisation of biomass samples for utilisation in thermal processes.
The moisture content of biomass is a crucial efficiency parameter when biomass combustion is the main consideration,
but less important in some lignocellulose hydrolysis technologies. Its determination is also necessary in most carbohydrate analytical procedures and it has a
huge influence on the near infrared spectra of lignocellulosic materials.
Water is generally held in biomass in two ways - either as a free liquid and vapour that is contained in the cell cavities, or as a molecule that is bound within the cell walls.
Moisture content tends to vary widely with biomass species, age, geographic locations and genetic differences. It also varies between different anatomical fractions of the same plant and throughout the year.
Water is generally held in biomass in two ways - either as a free liquid and vapour that is contained in the cell cavities, or as a molecule that is bound within the cell walls.
Moisture content tends to vary widely with biomass species, age, geographic locations and genetic differences. It also varies between different anatomical fractions of the same plant and throughout the year.
The moisture content of biomass can either be measured on a wet or dry basis. The wet-basis expresses the ratio of moisture mass to the total mass of the substance
whilst the dry basis expresses the ratio of the moisture mass to the mass of dry matter. All Celignis moisture data are reported using the wet-basis. Many of our thermal analysis
packages (e.g. P40) report thermal properties, such as the ash content and calorific value, on both the dry and as-received
(i.e. including moisture) bases.
These conversions to different bases are undertaken according to standard method EN 15296:2011.
Similarly, our data regarding the biomethane potential of anaerobic digestion feedstocks are reported on as-received as well as dry bases.
Similarly, our data regarding the biomethane potential of anaerobic digestion feedstocks are reported on as-received as well as dry bases.
Request a QuoteMoisture Content
Ash is generally considered to be the residue remaining after the material has been
incinerated. It therefore has no energy value and, being made up of the inorganic elements in the biomass, is of no direct
value in hydrolysis technologies. High ash-contents can cause problems in many
thermochemical processes
(e.g. pyrolysis, gasification, and combustion).
Ash content can vary greatly between plant species, and is generally higher in agricultural residues. The ash present in plants will depend on their stage of growth, the time of year, and their location. The leaching of stored biomass may reduce the level of inorganics in some instances.
Ash content can vary greatly between plant species, and is generally higher in agricultural residues. The ash present in plants will depend on their stage of growth, the time of year, and their location. The leaching of stored biomass may reduce the level of inorganics in some instances.
The major cations present in ashes from lignocellulosic materials are Calcium, Potassium and
Magnesium. Other elements such as Manganese, Sulphur and
Phosphorus are present in minor amounts.
Trace constituents (such as Al, Fe, Zn,
Cu, Ti, Pb,
Ni, V, Co,
Ag and Mo) are also found in many substrates.
The anions that are usually present are Chloride, Carbonate, Sulphate and Silicate.
With waste feedstocks (municipal solid wastes in particular), ashes are often more abundant and more diverse.
At Celignis with our elemental analysis methods we are able to characerise a wide range of major and minor analytes present in biomass ash. Click here to see our relevant analysis packages.
At Celignis with our elemental analysis methods we are able to characerise a wide range of major and minor analytes present in biomass ash. Click here to see our relevant analysis packages.
Request a QuoteAsh Content
Ash melting, also known as ash fusion and ash softening, can lead to slagging,
fouling and corrosion in boilers which may reduce conversion efficiency.
We can determine the ash melting behaviour of biomass using our Carbolite CAF G5 BIO ash melting furnace.
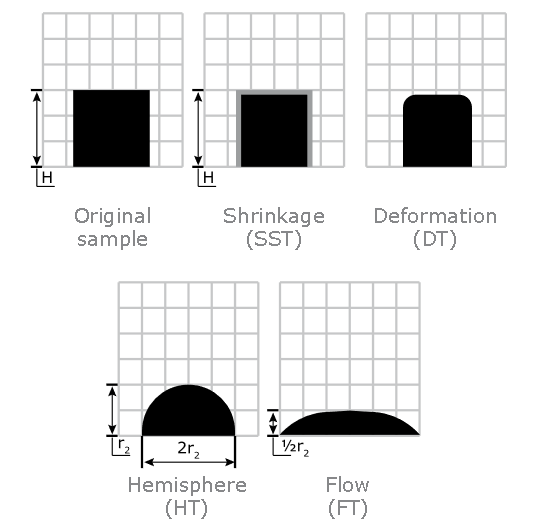
It can record the following temperatures:
Ash Shrinkage Starting Temperature (SST) - This occurs when the area of the test piece of biomass ash falls below 95% of the original test piece area.
Ash Deformation Temperature (DT) - The temperature at which the first signs of rounding of the edges of the test piece occurs due to melting.
Ash Hemisphere Temperature (HT) - When the test piece of biomass ash forms a hemisphere (i.e. the height becomes equal to half the base diameter).
Ash Flow Temperature (FT) - The temperature at which the biomass ash is spread out over the supporting tile in a layer, the height of which is half of the test piece at the hemisphere temperature.
The ultimate analysis of biomass will provide the mass concentrations of the major elements (carbon, oxygen,
hydrogen, nitrogen and sulphur) in the sample.
These concentrations will depend on the chemical composition of the biomass sample.
Lignocellulosic biomass is mainly composed of carbohydrates (in the form of the polysaccharides cellulose and hemicellulose) and lignin. Carbohydrates, having an elemental composition of (CH2O)n, will have a generally uniform carbon content of 40%, which is lower than many other biomass mass constituents such as lignin which has an average carbon content of approximately 60-65%.
Typically carbon, hydrogen, nitrogen, and sulphur can be determined in the same analytical method and the oxygen content can then be calculated as the remaining mass after the ash content has been considered.
We use an Elementar MACRO Cube unit for the ultimate analysis of samples. It allows for large sample sizes (e.g. around 40mg) to be used than many other elemental analysers, allowing us to improve the precision in the analysis of biomass and waste samples.
Lignocellulosic biomass is mainly composed of carbohydrates (in the form of the polysaccharides cellulose and hemicellulose) and lignin. Carbohydrates, having an elemental composition of (CH2O)n, will have a generally uniform carbon content of 40%, which is lower than many other biomass mass constituents such as lignin which has an average carbon content of approximately 60-65%.
Typically carbon, hydrogen, nitrogen, and sulphur can be determined in the same analytical method and the oxygen content can then be calculated as the remaining mass after the ash content has been considered.
We use an Elementar MACRO Cube unit for the ultimate analysis of samples. It allows for large sample sizes (e.g. around 40mg) to be used than many other elemental analysers, allowing us to improve the precision in the analysis of biomass and waste samples.
Request a QuoteUltimate Analysis
The heating value is currently one of the most important properties of biomass given the predominance of combustion facilities over
other biomass valorisation technologies.
There are several units of measurement. The caloric, or higher heating value (HHV), is independent of moisture content and reliant on the chemical composition of the material. A linear relationship exists between the heat of combustion and the carbon content of the substrate while oxygen, nitrogen and inorganic elements tend to reduce the value.
The Lower Heating Value (LHV), or effective heating value, is perhaps more relevant than the HHV in practical operations. It considers not only the energy required to vaporise the moisture of the biomass but also that necessary to vaporise the water generated when the hydrogen and oxygen elements of the biomass combine. Hydrogen content then becomes a reducing factor in the heating value.
There are several units of measurement. The caloric, or higher heating value (HHV), is independent of moisture content and reliant on the chemical composition of the material. A linear relationship exists between the heat of combustion and the carbon content of the substrate while oxygen, nitrogen and inorganic elements tend to reduce the value.
The Lower Heating Value (LHV), or effective heating value, is perhaps more relevant than the HHV in practical operations. It considers not only the energy required to vaporise the moisture of the biomass but also that necessary to vaporise the water generated when the hydrogen and oxygen elements of the biomass combine. Hydrogen content then becomes a reducing factor in the heating value.
The heating value of samples can be determined directly, by combusting the sample in a bomb calorimeter,
or it can be calculated based on elemental composition. The heating value can be reported on a dry or wet basis.
The wet-basis value considers the moisture content of the biomass and reduces the heating value to incorporate the latent heat of condensation.
At Celignis we use a Parr 6200 bomb calorimeter to directly determine the gross calorific value (higher heating value) of samples. The data from ultimate analysis are then used to calculate the net calorific value (lower heating value).
At Celignis we use a Parr 6200 bomb calorimeter to directly determine the gross calorific value (higher heating value) of samples. The data from ultimate analysis are then used to calculate the net calorific value (lower heating value).
Request a QuoteHeating Value